Katie Bird is a Senior Enforcement Officer at the Office for Product Safety and Standards (OPSS), part of the UK Government’s Department for Business, Energy and Industrial Strategy (BEIS) and the UK’s competent national authority for Access and Benefit Sharing (ABS).
Zoological Biobanks for Species Conservation
Worldwide populations of vertebrates have already declined by 60% between 1970 and 2014 [1]. Finding solutions to species recovery often depends on collecting DNA samples, which can reveal valuable information about details ranging from inbreeding and population history to natural selection and large-scale threats such as habitat destruction and illegal wildlife trade [2]. Species extinctions and declining populations are not just undesirable but have a direct impact on the quality of human lives. Biodiversity loss can cause an increase in infectious disease transmission in humans and animals [3] as well as rob humans of potential medical advances by providing less opportunity to study novel compounds or mechanisms found in animals [4]. Without a significant increase in conservation efforts, species extinction and declines are likely to continue at an alarming rate [5].
Recognising the importance of Biodiversity
In recognition of the importance and value of biodiversity, the Convention on Biological Diversity (CBD) came into effect in 1993 with three main objectives:
(1) to conserve biodiversity;
(2) to ensure the sustainable use of biodiversity; and
(3) to ensure the fair and equitable sharing of benefits arising from the utilisation of genetic resources.
The Nagoya Protocol on Access to Genetic Resources and the Fair and Equitable Sharing of Benefits Arising from their Utilisation, or ‘Nagoya Protocol’ for short, came into force in October 2014, creating a legal framework for the third objective’s effective implementation whilst considering the other two objectives in practice. “Genetic resources” are defined (in the CBD) as “genetic material of actual or potential value”; in turn, “genetic material” means “any material of plant, animal, microbial or other origin containing functional units of heredity”, but not including human genetic material. “Utilisation of genetic resources” is defined in the Nagoya Protocol as “to conduct research and development (R&D) on the genetic and/or biochemical composition of genetic resources…” [6].
EU Exit
The UK left the EU on 31st January 2020 and the withdrawal agreement has taken effect. From that date, the UK has entered into an “Transition Period” until the end of 2020. During the Transition Period, EU Regulation 511/2014 on compliance measures for the Nagoya Protocol will remain in effect and due diligence declarations will still be submitted through the EU portal DECLARE. More information will be updated on the website www.gov.uk/abs when it is available.
How to Comply
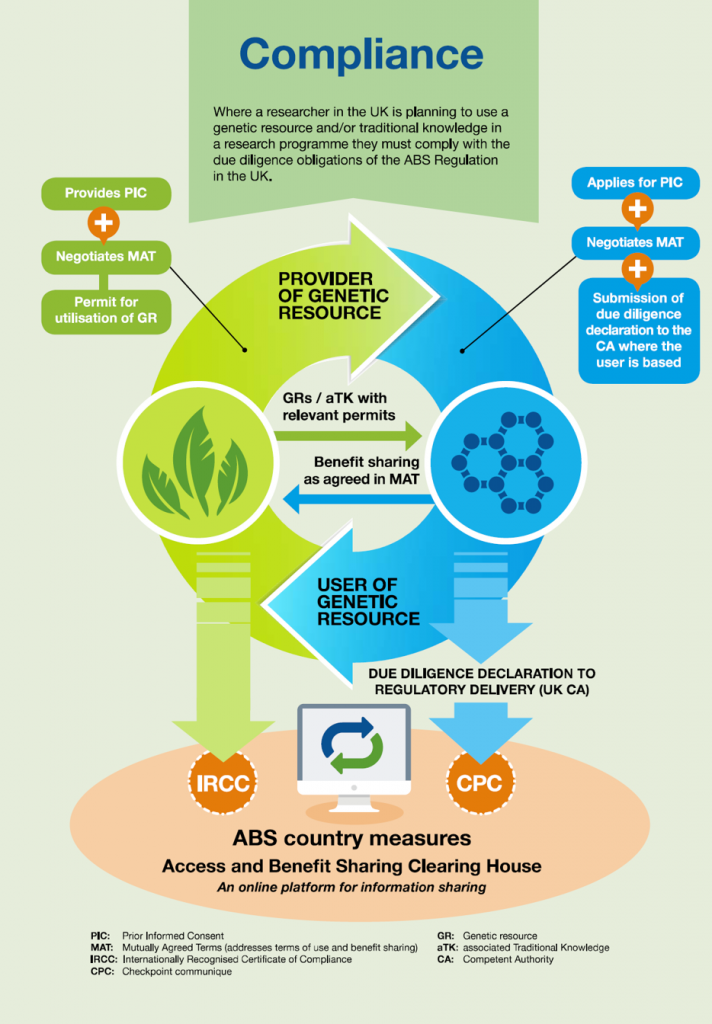
In the EU, compliance measures for the Nagoya Protocol are contained in EU Regulation No 511/2014 (the European ABS Regulation) and are transposed in to UK law through UK Statutory Instrument, No. 821. Those conducting R&D on genetic resources or associated traditional knowledge are legally required to exercise due diligence to seek and comply with access legislation in the provider country. Depending on the national measures that apply, the user may need to demonstrate prior informed consent (PIC) to access the genetic resource and negotiate mutually agreed terms (MAT) for undertaking R&D that provides for benefit sharing opportunities. The EU ABS Regulation applies to genetic resources from countries party to the Nagoya Protocol that have access legislation in place and were accessed after 12 October 2014. Compliance with the EU regulations includes exercising due diligence to ensure the material was accessed in accordance with local laws or regulations, seeking, keeping and transferring information related to the genetic resource and submitting due diligence declarations containing this information at various checkpoints (Figure 1).
If you are creating new knowledge about the characteristics of the genetic resource then you have the potential to be in scope of the EU ABS Regulation and are therefore required to exercise due diligence to seek and comply with access legislation in the provider country [7]. For example, the creation of a genetically modified animal, plant, or microorganism containing a gene from another species would be considered in scope of the EU ABS Regulation, however the mere planting and harvesting of seeds is out of scope.
Access measures vary from country to country and the best source of information for this is the Access and Benefit Sharing Clearing House (ABS-CH). Here, you can find if a country is a party to the Nagoya Protocol, contact details for the National Focal Points (NFP) and Competent National Authorities (CNAs) of most countries, as well as any access legislation in place. The aim of access legislation is to ensure that the conservation and sustainable use of biodiversity is considered prior to access and to prevent unfair exploitation of a country’s genetic resources or traditional knowledge. It is important to remember that even if a genetic resource does not fall within the scope of the EU ABS Regulation, the provider country may still have access legislation that the user must comply with.
Responsibilities of Collections
In the EU regulations, the maintenance and management activities associated with collections are out of scope, including quality/phytopathology checks, and verification of material upon acceptance. Therefore, collections that do not conduct R&D as mentioned above do not have the same responsibilities of compliance under the EU ABS compliance measures. There may be laws in the country of origin that the collection will need to follow in order to access materials, but the collection would not be under any obligation to comply with the EU regulation.
Collections can apply to become registered collections within the EU. The registration process is detailed in the EU implementing regulation. When a collection becomes a registered collection, they then have a legal obligation to supply genetic materials only with documentation providing evidence that the material and associated information were accessed in accordance with applicable ABS legislation, and mutually agreed terms where applicable. Users obtaining material from a registered collection are considered to have exercised due diligence regarding seeking the information on that genetic resource [8].
Conclusion
If a user performs R&D on genetic materials, that were accessed after 12 October 2014, from a country that is a party to the Nagoya Protocol and has access measures in place, they have the potential to be in scope of the ABS compliance measures in force in the UK. This means they need to exercise due diligence to ensure it was collected in accordance with applicable legislation in the providing country.
The Departments for Environment, Food and Rural Affairs (DEFRA) and Business Energy and Industrial Strategy (BEIS) are responsible for implementing the Nagoya Protocol in the UK. The Office for Product Safety and Standards (OPSS), part of BEIS, acts as the CNA and works directly with business, research organisations and other institutions to support understanding and compliance. For any further information please visit gov.uk/abs or email us at opss.enquiries@beis.gov.uk
References
[1] WWF (2019) Living Planet Report 2018. Aiming Higher. WWF International, Gland, Switzerland.
[2] Stanford University. (2019, April 10). Scant amounts of DNA reveal conservation clues. ScienceDaily. Retrieved January 5, 2020 from www.sciencedaily.com/releases/2019/04/190410083108.htm
[3] Humans are more at risk from diseases as biodiversity disappears. https://blogs.scientificamerican.com/extinction-countdown/humans-are-more-at-risk-from-diseases-as-biodiversity-disappears/
[4] How Do the Extinctions of Other Creatures Affect Humans Directly? https://sciencing.com/extinctions-other-creatures-affect-humans-directly-20692.html
[5] Valk, T.V. (2018). From conservation genetics to conservation genomics using minimal-invasive sampling methods.
[6] Convention on Biological Diversity. Text of the Nagoya Protocol https://www.cbd.int/abs/text/
[7] EU Guidance document https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.C_.2016.313.01.0001.01.ENG&toc=OJ:C:2016:313:TOC
[8] EU Guidance document https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.C_.2016.313.01.0001.01.ENG&toc=OJ:C:2016:313:TOC
